Health

Identification and quantification of biomarkers
Tissue engineering
Microinjected health products
Telemedicine or e-Health
Health industry equipment
Tekniker´s offer for the health sector encompasses the following areas:
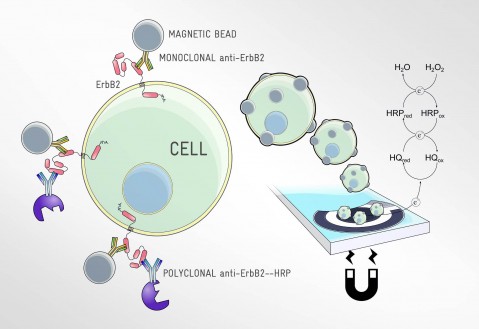
IDENTIFICATION AND QUANTIFICATION OF BIOMARKERS
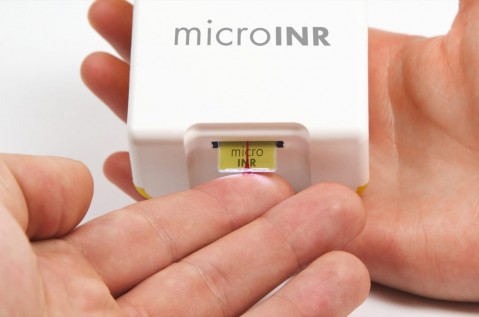
- Development of easy-to-use and low-cost devices for outpatient care. Portable diagnostic and measuring devices featuring embedded systems, measuring algorithms and communications that comply with health sector standards and certification requirements.
- Development of devices based on biomarker control and monitoring for body fluids (liquid biopsies) and customised medicine.
- Rapid, low concentration detection using micro and nanotechnology capabilities.
- Tailor-made multiplexed detection capabilities for specific biomarkers.
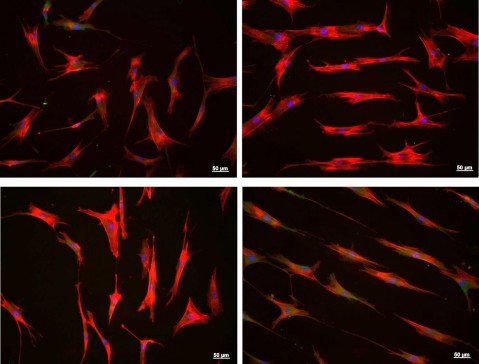
TISSUE ENGINEERING
- Development of biofunctional surfaces for companies developing cell therapies and organisations such as implant manufacturers that require surfaces of this kind.
- Development of 2D and 3D extracellular matrices (scaffolds) in a variety of materials.
- Specific geometric designs to favour cell differentiation and proliferation.
- Development of coatings and/or textures for biofunctional surfaces to speed up osseointegration or form biocide layers with antibacterial activity.
MICROINJECTED HEALTH PRODUCTS
- Development of health products using biocompatible and/or biodegradable microinjected polymers.
- The production of prototypes to support the design phase and feasibility.
- Injection simulations to guarantee that all cavities are filled.
- Design, manufacture and fine-tuning of moulds.
- Manufacture of pre-series prior to the commercial launch itself.
- Process and product monitoring to achieve "zero defect" manufacturing”.
TELEMEDICINE OR E-HEALTH
- Development of telemedicine systems (support for health self-management, telemonitoring and telerehabiltation adapted to the specific needs of individual patients).
- Development of systems for chronic and/or elderly patients so they properly manage their disease. These developments address aspects such as monitoring medical parameters and lifestyle and training on how to manage risk situations linked to mood or lifestyle changes, etc. These systems are incorporated to those already used by health care providers in accordance with the standards set for this purpose.
- Development of smart decision making support systems such as natural language processing, semantic technologies and automatic learning to assist both patients and health care professionals in the decision making process in the diagnostic and treatment phases based on the patient´s profile and available scientific evidence.
HEALTH INDUSTRY EQUIPMENT
- Development of service robots equipped with autonomous navigation capabilities to assist other persons and robots and which can provide guidance at hospitals or public environments and perform logistic activities at hospitals.
- Development of assisting robots to help elderly people or individuals with functional constraints at home or nursing facilities.
- Drafting of specifications to transfer laboratory processes to repeatable and reproducible processes.
- Design and development of equipment meeting criteria on good laboratory practices (GLP).